The history of ECG247
The ECG247 heart sensor represents many years of research and technology development in Norway.
The traditional equipment for assessment of heart rhythm disorders was developed in the 1960s. It is cumbersome to use, requires physical attendance at the hospital and can be used for a few consecutive days.
Professor Rune Fensli at the Center of e-health at the University of Agder in Grimstad, Norway sought for many years to develop a new and better solution for detecting heart rhythm disorders.

Cardiologist PhD Jarle Jortveit and professor Rune Fensli. Photo: Bård Larsen, Grimstad Adressetidende
The prototype of a new wireless smart heart sensor was developed in collaboration with the technology companies Jetro and Egde in Grimstad, Norway. Important requirements for the new sensor were ease of use, high diagnostic accuracy and a possibility of long continuous heart rate monitoring also during normal activities such as exercise and work. The sensor also had to be robust, reusable and inexpensive.
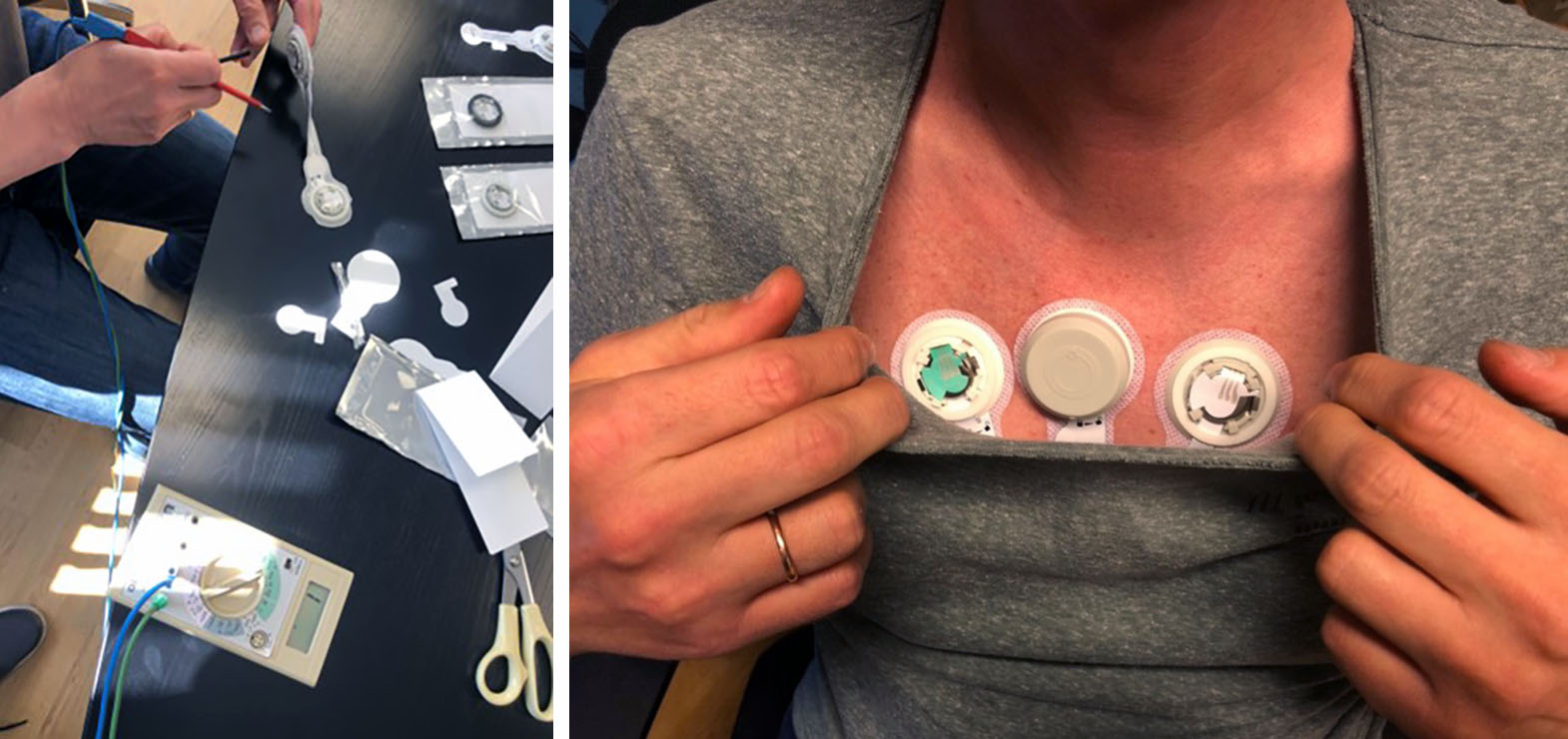
Development and testing of ECG247 at Jetro AS, Grimstad, Norway
ECG247 is manufactured at Kitron in Arendal and Verktøymakeren in Lillesand, Norway. The production, which is completely robotic, is subject to strict quality requirements at all stages.
Robotproduksjon av ECG247 sensorer hos Kitron AS, Arendal
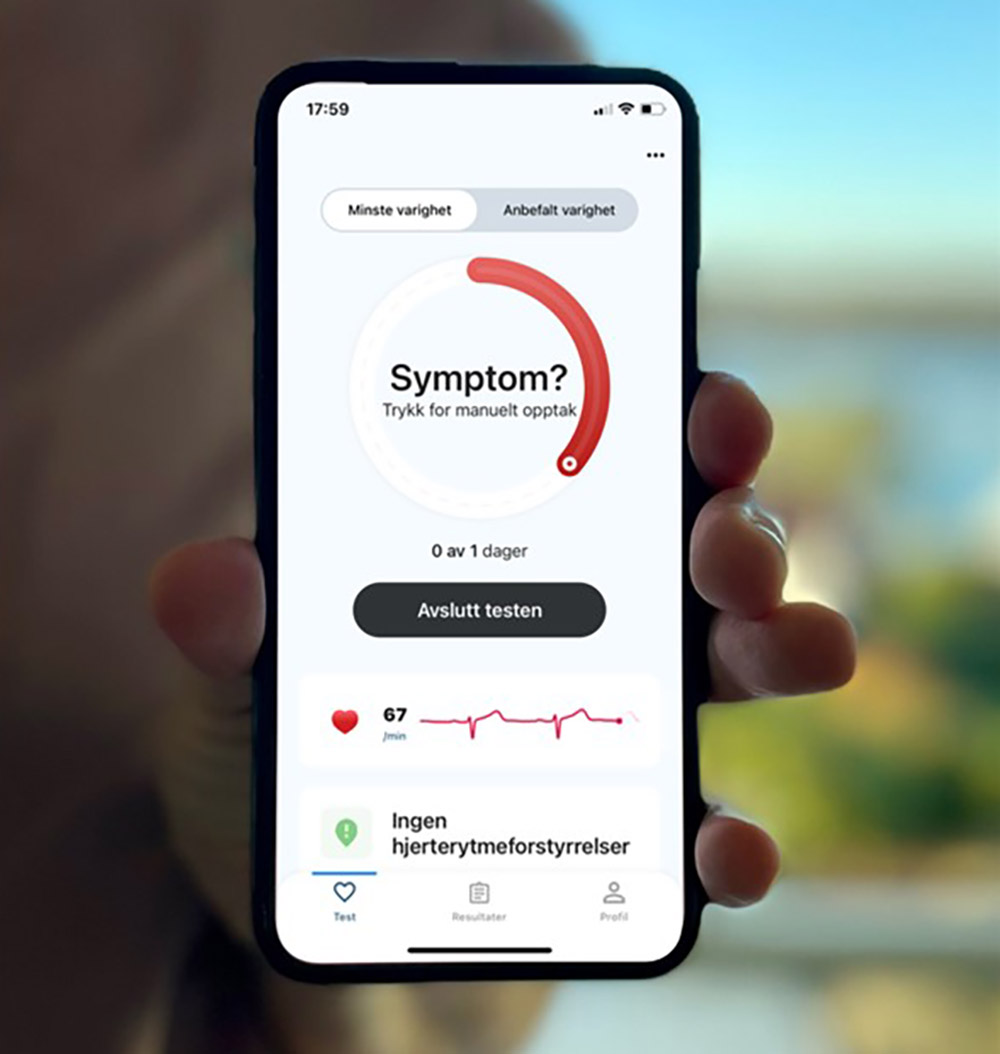
ECG247 app
Safety and accuracy are important for medical equipment. ECG247 was certified in the EU in 2020 according to European standards.
The ECG247 app and the ECG247 web portal communicate with the heart sensor to display the results in a simple way. Both services are developed and maintained by Apphuset in Bergen, Norway.
ECG247 is developed for clinical research (e.g. screening for atrial fibrillation), for use in the professional health care system and for self-testing/consumer-use.
ECG247 is in routine clinical use at many GPs and hospitals throughout Norway and other countries in Europe and Asia.

CEO of Appsens AS Tord Ytterdahl signs a cooperation agreement with the hospital chain GVM in Italy



